on ABIVAX (EPA:ABVX)
Abivax presents the results of the ABTECT trial
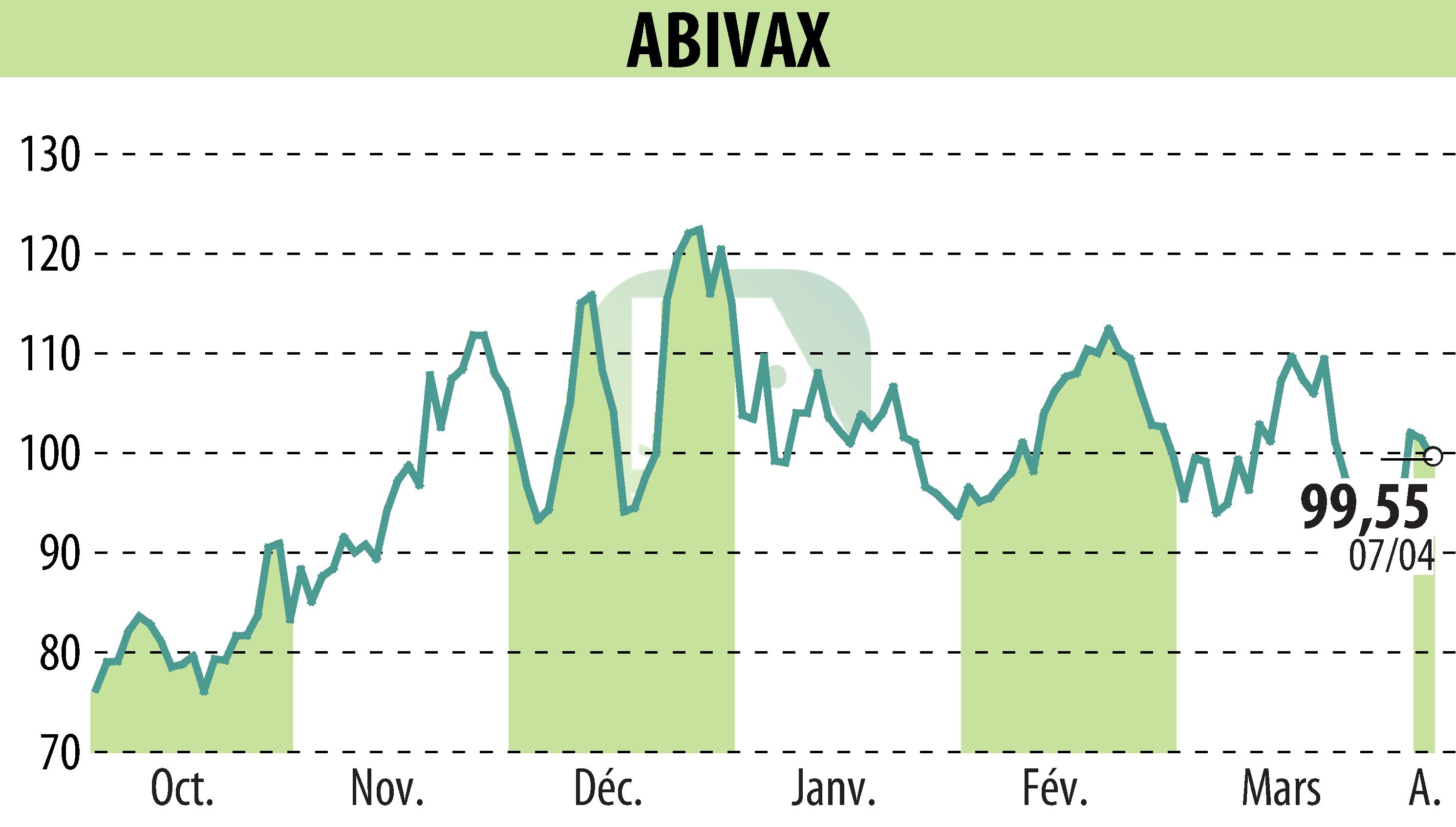
Abivax unveiled the results of its Phase 3 ABTECT trials at UEG Week in Berlin, highlighting obefazimod for the treatment of moderate to severe active ulcerative colitis. Administered at 50 mg daily, it achieved a clinically corrected remission rate of 16.4% (p < 0.0001) at week 8.
The study, conducted on 1,272 patients, included a refractory population, with 47% of participants having not responded to previous advanced treatments. Obefazimod demonstrated good tolerability, with side effects limited primarily to headaches and nausea. No new safety signals were observed.
Abivax will hold a conference to discuss these results on October 6. This presentation highlights the potential of Obefazimod as a new oral treatment in an area of significant unmet need.
R. P.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all ABIVAX news
