on Nanohale AG (ETR:FYB)
Formycon Secures Licensing for Biosimilar in Europe and Beyond
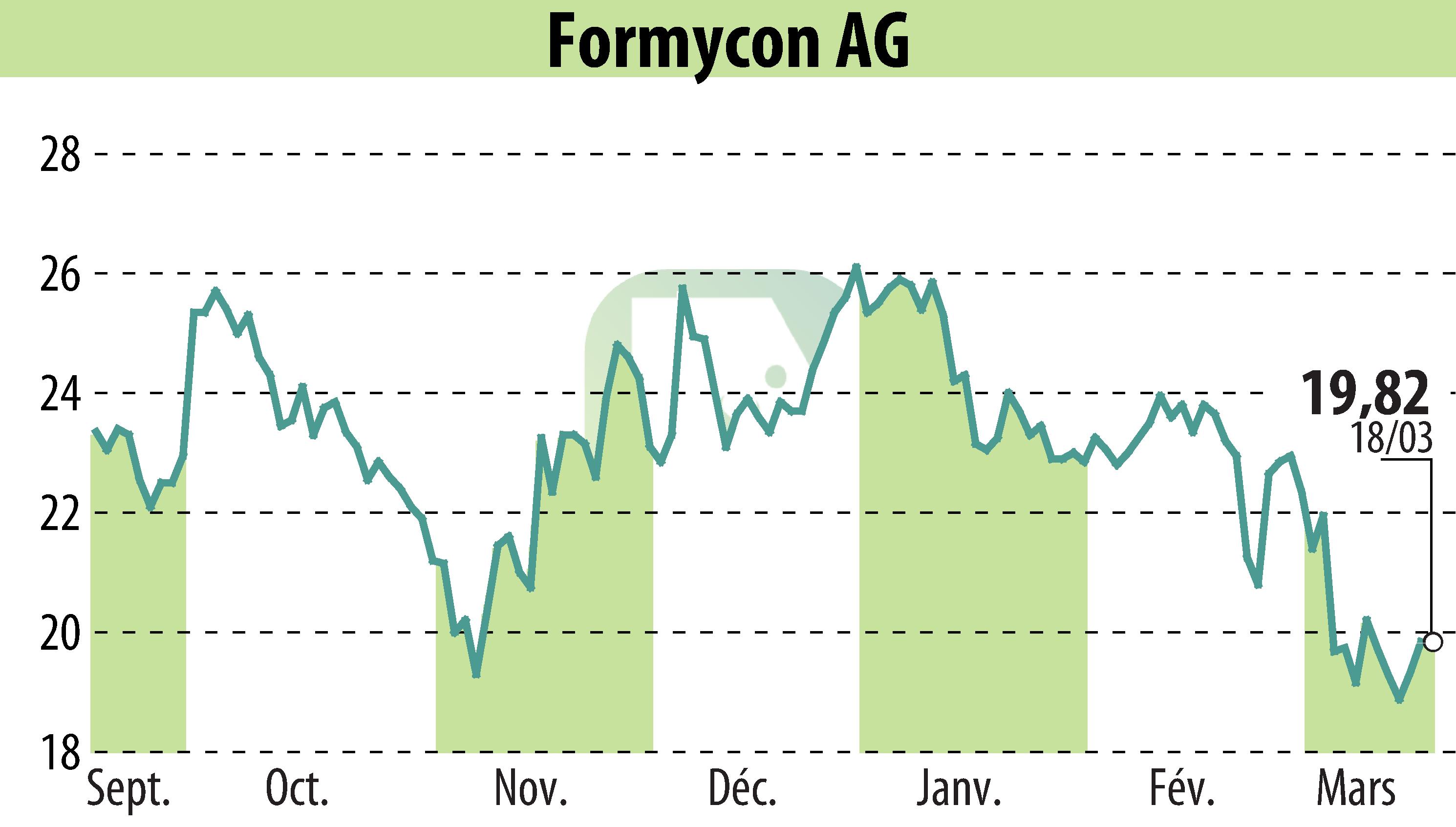
Formycon AG has finalized a settlement and licensing agreement with Regeneron Pharmaceuticals and Bayer Healthcare LLC for its biosimilar FYB203, also known as AHZANTIVE® and Baiama®. This agreement allows the release of these biosimilars in Europe and key markets in Latin America and the Asia-Pacific region starting in May 2026. This follows the October 2025 securing of the U.S. license date, planned for Q4 2026.
AHZANTIVE® and Baiama® target neovascular age-related macular degeneration and other serious retinal diseases, using aflibercept to inhibit the growth of damaging blood vessels in the retina. The settlement resolves all patent litigations, facilitating market access for commercialization partners. For Formycon, this represents a strategic expansion and opportunity for sustainable success in international markets, offering a cost-effective treatment option for retinal diseases.
R. H.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all Nanohale AG news
