on INNATE PHARMA (EPA:IPH)
Innate Pharma presents interim results from the MATISSE trial at the AACR congress
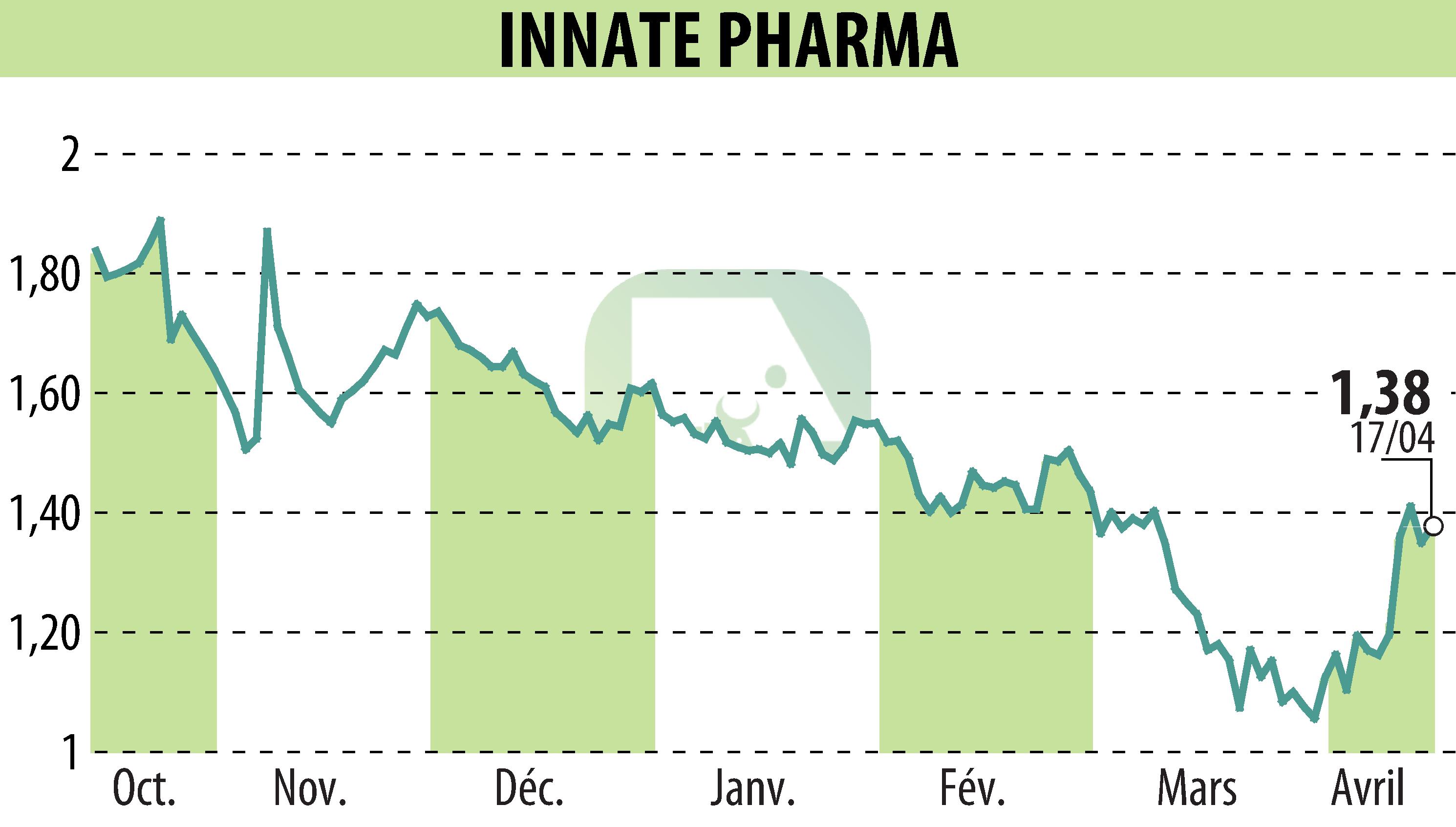
Innate Pharma announced the presentation of interim results from the Phase 2 MATISSE trial at the AACR 2026 Annual Meeting. This trial evaluates the monoclonal antibody IPH5201, combined with durvalumab and chemotherapy, in the treatment of operable non-small cell lung cancer.
The study showed encouraging results, with complete pathological response rates of 37.7% and 50% in patients whose tumors expressed PD-L1 ≥1% and PD-L1 ≥50%, respectively. These results, based on 40 patients, support continued recruitment for tumors expressing PD-L1 ≥1%.
By adopting a dual inhibition of the CD39 and PD-L1 pathways, combined with chemotherapy, Innate Pharma hopes to improve antitumor immune responses and clinical outcomes in early-stage lung cancer.
R. H.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all INNATE PHARMA news
