on INNATE PHARMA (EPA:IPH)
Innate Pharma Reveals Promising Interim Results in MATISSE Phase 2 Study
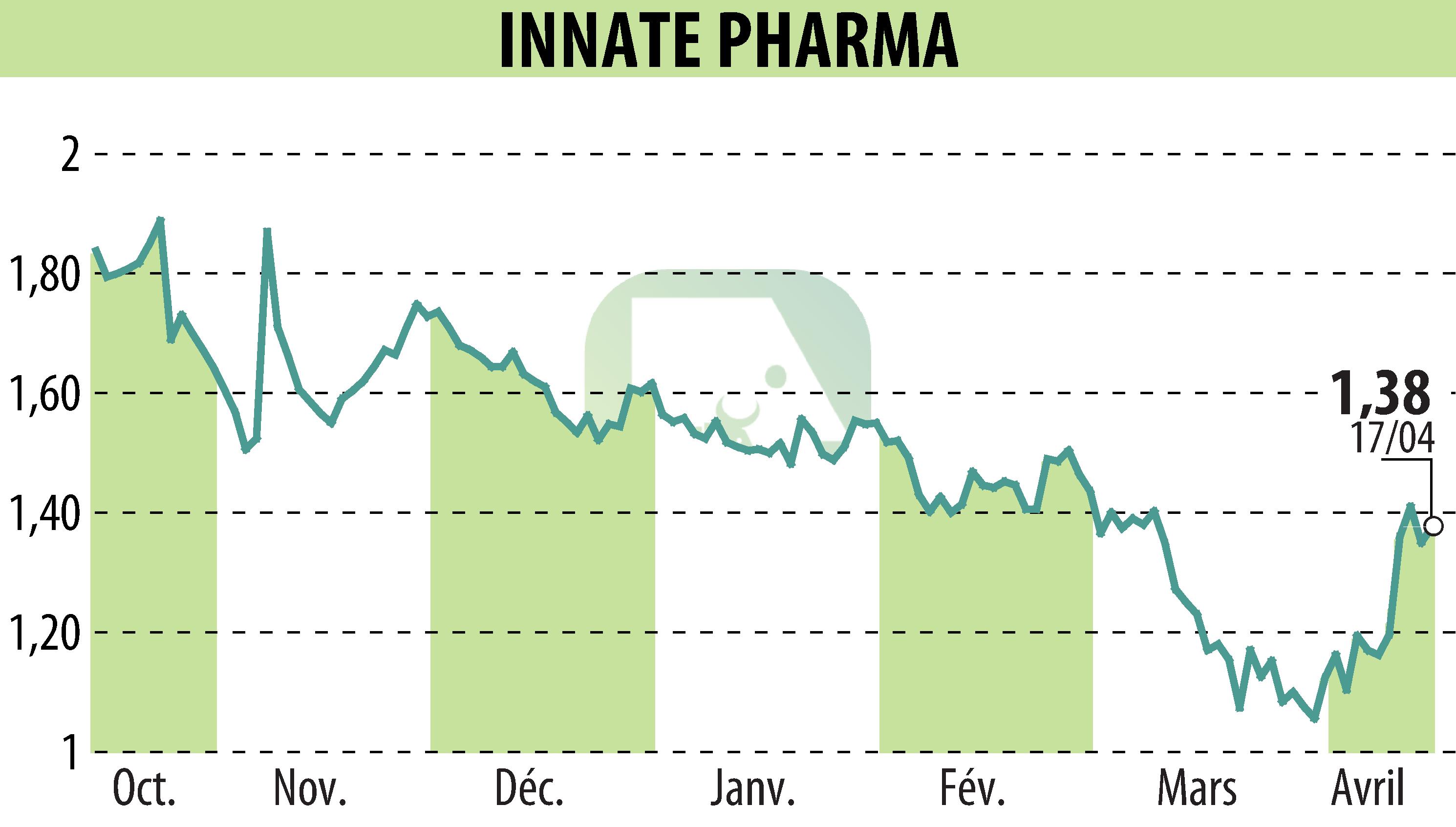
Innate Pharma has disclosed interim results from its MATISSE Phase 2 study evaluating IPH5201. This monoclonal antibody, combined with durvalumab and chemotherapy, shows encouraging early results in treating resectable non-small cell lung cancer (NSCLC). Presented at the AACR Annual Meeting 2026, these findings highlight enhanced anti-tumor responses.
The trial involved 40 patients, revealing higher pathological complete response rates. Notably, a 35.7% and 50% response rate was observed in patients with PD-L1 ≥1% and PD-L1 ≥50% tumors, respectively. This suggests a potential clinical advancement for PD-L1 positive early-stage lung cancer patients.
Dr. Sonia Quaratino, Chief Medical Officer, emphasized the potential of IPH5201 in disrupting the adenosine pathway to boost immune responses in these patients. This ongoing study continues to recruit to expand understanding.
R. E.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all INNATE PHARMA news
