on Jaguar Health, Inc. (NASDAQ:JAGX)
Jaguar Health Completes Crofelemer Trial for CID in Dogs
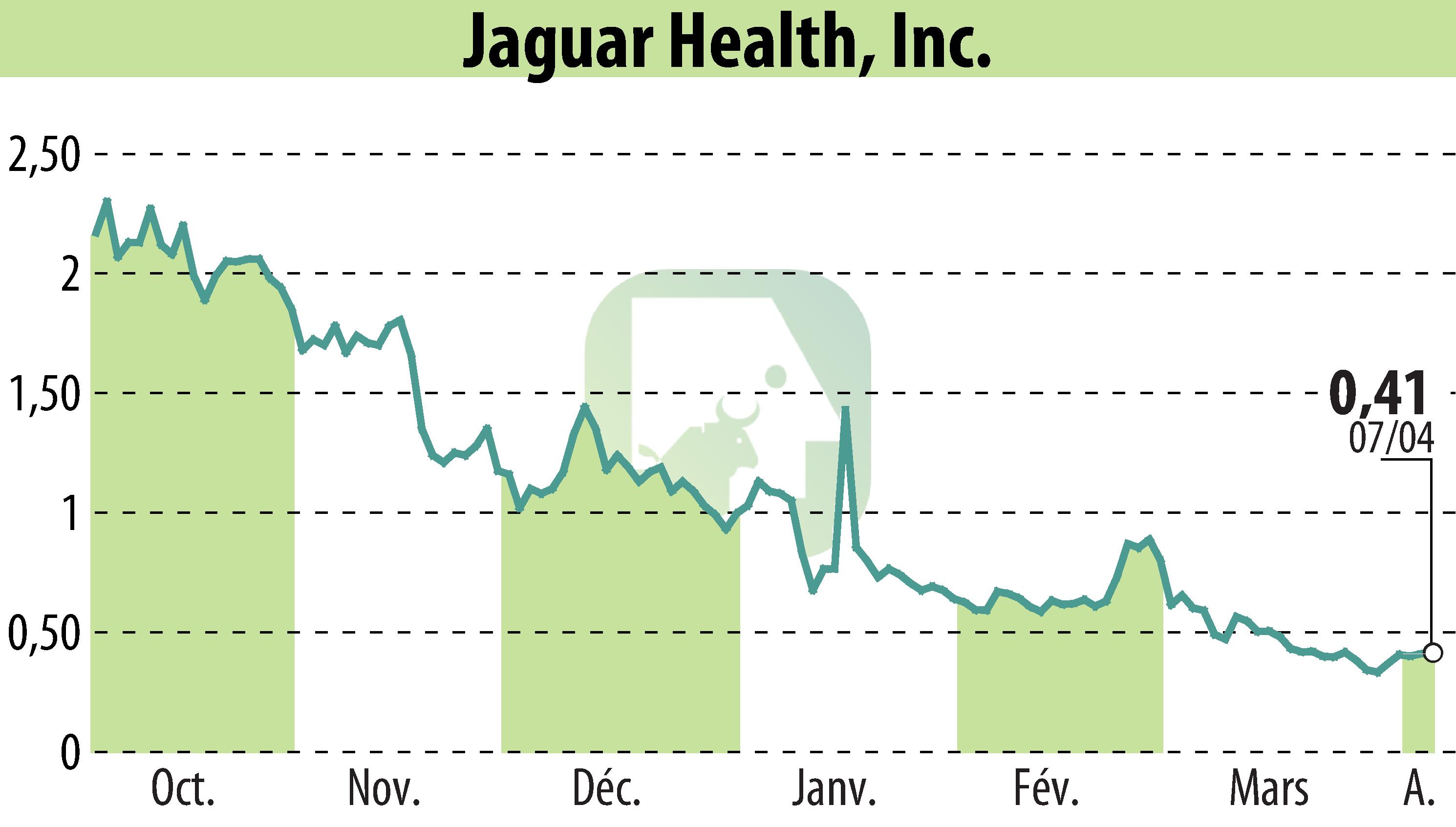
Jaguar Health, Inc. has completed its effectiveness trial of crofelemer delayed-release tablets for treating chemotherapy-induced diarrhea (CID) in dogs. Crofelemer, marketed as Canalevia-CA1, is conditionally approved for this use. The conditional approval expires on December 21, 2026.
The recent study enrolled dogs at veterinary oncology clinics across the U.S. Results are expected within a month and may support the approval process for the drug. Jaguar plans to submit the study results to the FDA in the second quarter of 2026.
A complementary study demonstrated crofelemer's effectiveness in reducing diarrhea severity in dogs treated with neratinib, a targeted cancer therapy. This study showed a statistically significant 30% reduction in diarrhea incidence and severity.
Crofelemer is not an antibiotic, minimizing concerns about antibiotic resistance. As more dogs undergo chemotherapy, effective management of side effects like diarrhea becomes increasingly vital.
R. E.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all Jaguar Health, Inc. news
