on ORPHAN SYNERGY EUROPE-PHARMA (EPA:OSE)
OSE Immunotherapeutics Celebrates FDA Orphan Drug Designation for Pegrizeprument
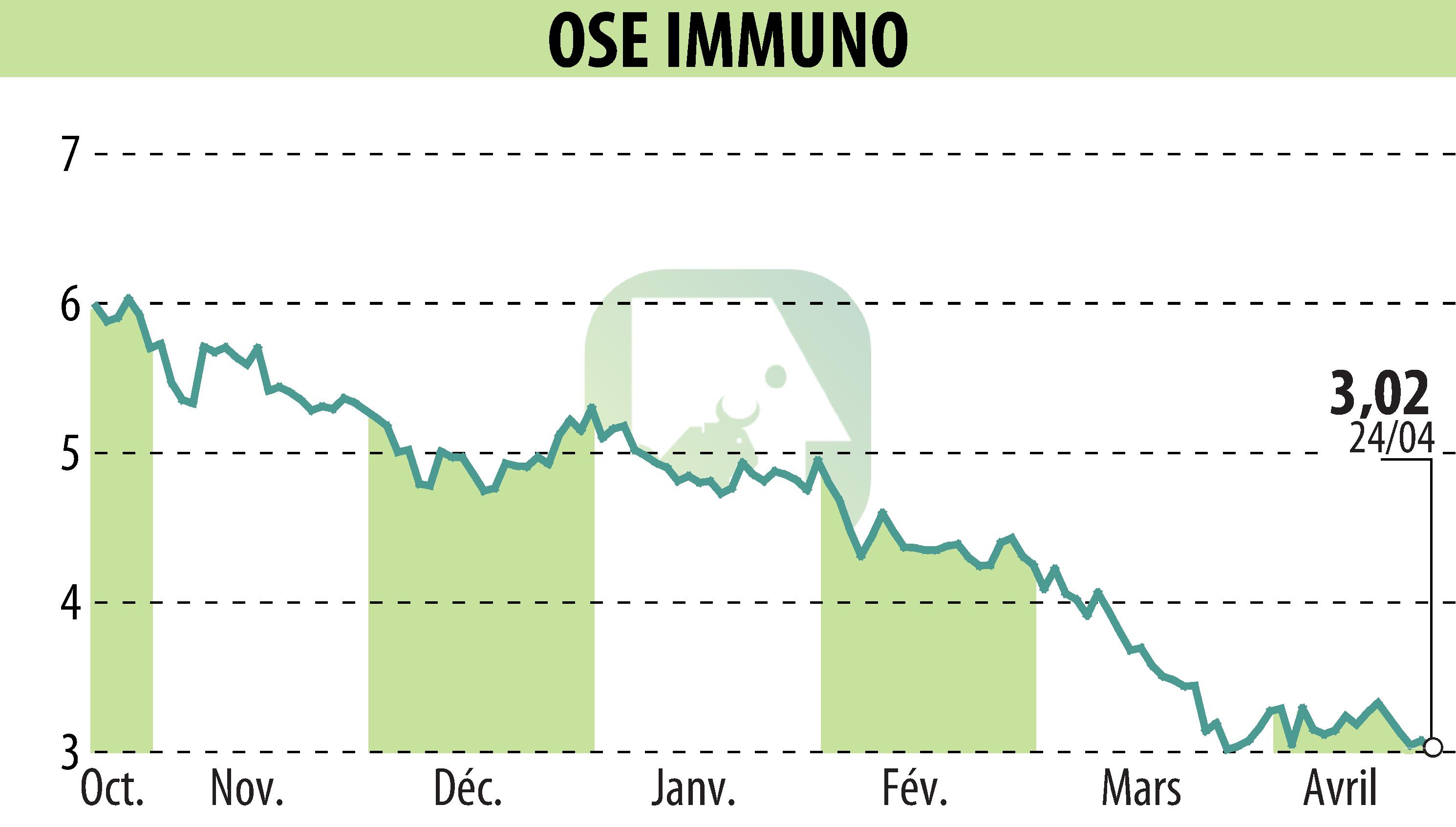
OSE Immunotherapeutics announced that the FDA has granted Orphan Drug Designation to Pegrizeprument (VEL-101) for preventing heart transplant rejection. This declaration follows a previous designation in January 2026 for liver transplant rejection.
Pegrizeprument, an investigational monoclonal antibody fragment, was licensed to Veloxis Pharmaceuticals in 2021. Veloxis handles global development and commercialization.
The FDA’s Orphan Drug Designation targets treatments for rare diseases affecting fewer than 200,000 US patients, aiming to promote drug development for underserved conditions.
Email addresses and contact details are available for investor and media relations, highlighting OSE’s commitment to transparency and engagement with stakeholders.
R. E.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all ORPHAN SYNERGY EUROPE-PHARMA news
