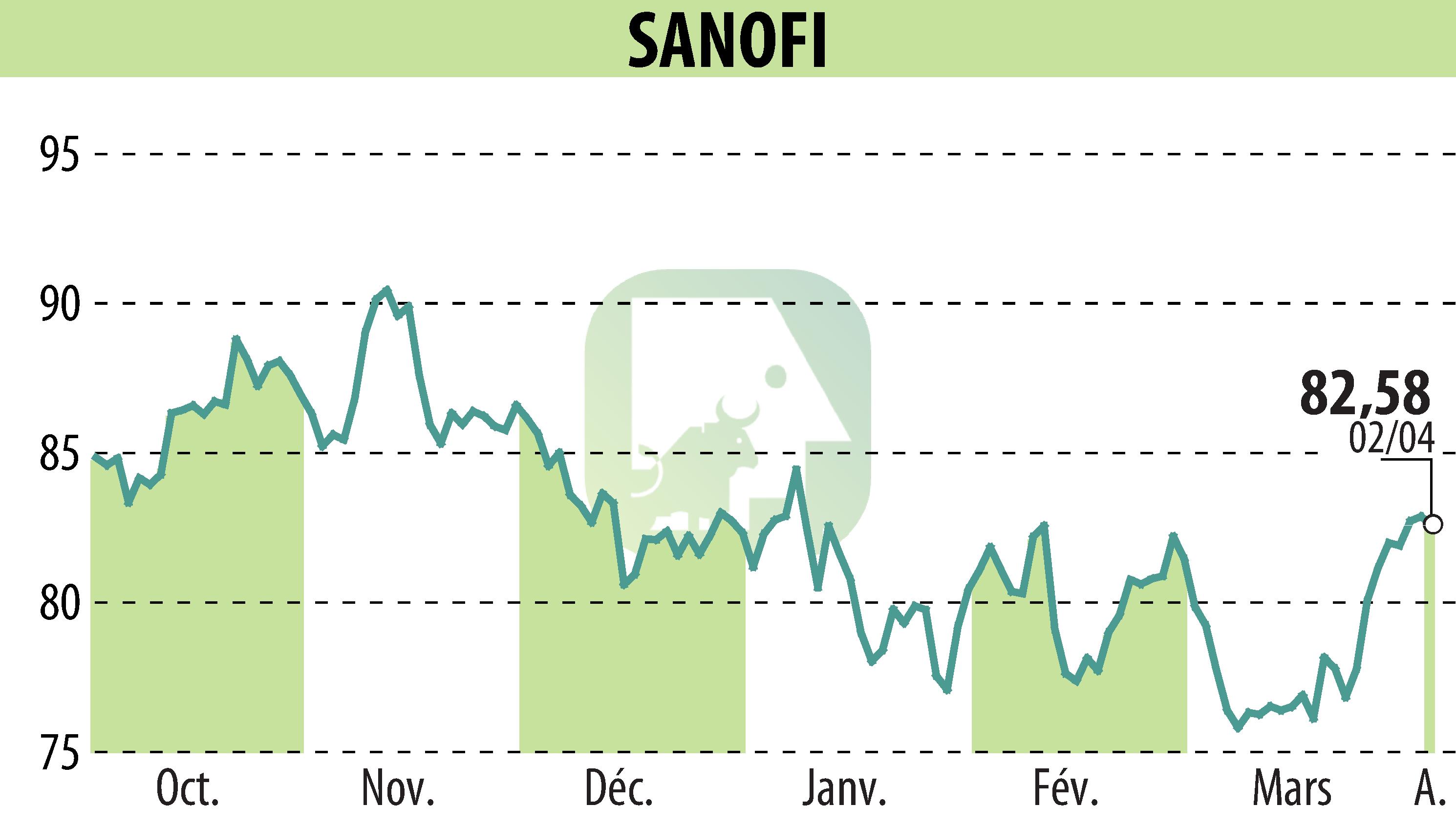
on SANOFI-AVENTIS (EPA:SAN)
Sanofi's Lunsekimig Shows Promise in Asthma and CRSwNP Studies
Sanofi's recent phase 2 studies reveal promising results for Lunsekimig in treating chronic respiratory conditions. The AIRCULES phase 2b trial successfully met its primary and secondary objectives in moderate-to-severe asthma, highlighting a significant reduction in exacerbations and lung function improvement. Similarly, the DUET phase 2a study achieved its targets in chronic rhinosinusitis with nasal polyps (CRSwNP), demonstrating positive changes in nasal polyps and congestion scores. However, the VELVET phase 2b study did not achieve its primary goal in atopic dermatitis.
Lunsekimig, a novel bispecific Nanobody® VHH, blocks TSLP and IL-13, key factors in respiratory inflammation. Across all studies, the drug was generally well tolerated. Detailed findings will be presented at future medical congresses. These developments emphasize Lunsekimig's potential role in managing respiratory diseases, although regulatory evaluations remain pending.
R. P.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all SANOFI-AVENTIS news
