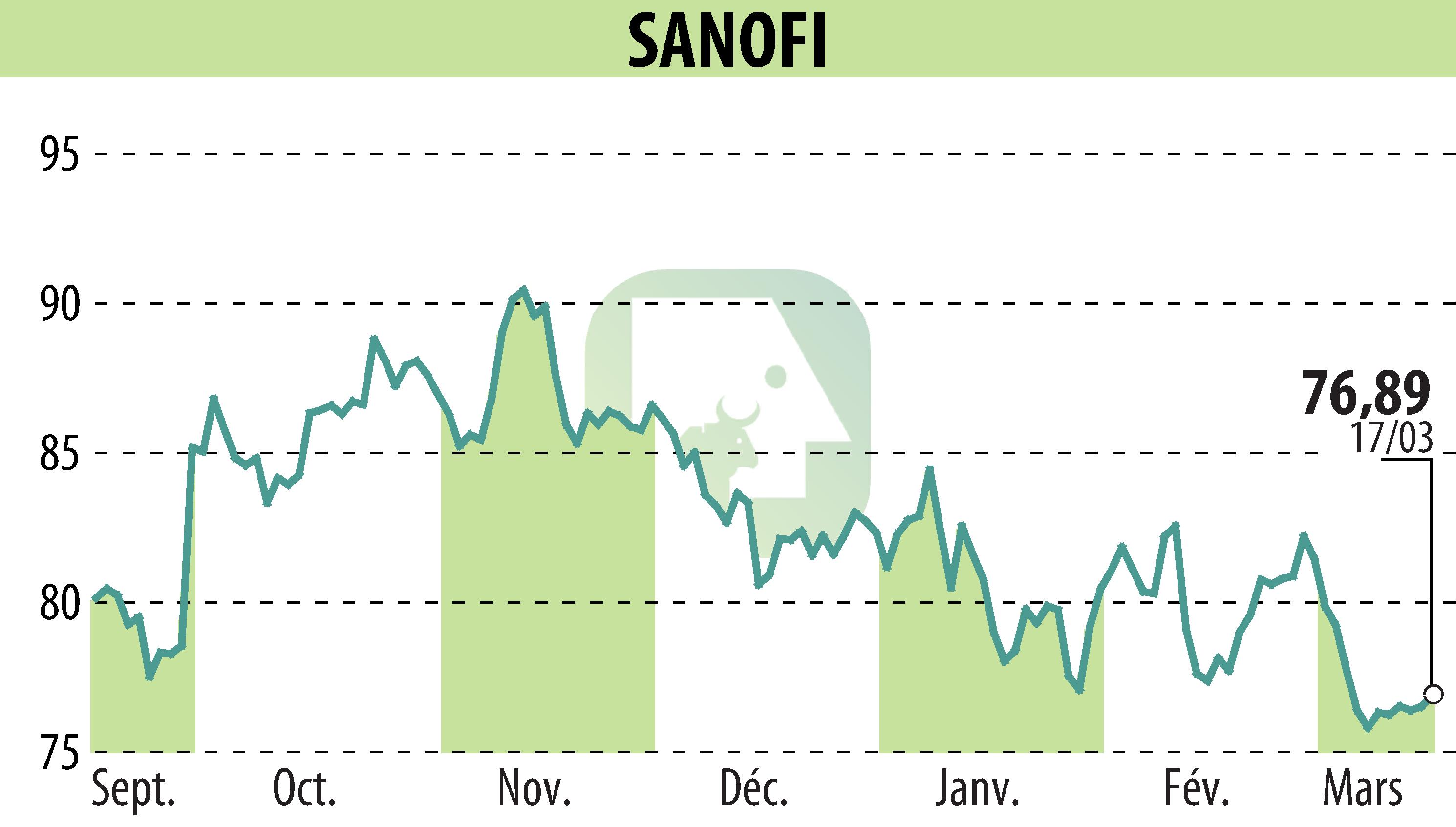
on SANOFI-AVENTIS (EPA:SAN)
Sanofi: Breakthrough Therapy Designation for venglustat in the United States
Sanofi announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Therapy designation to venglustat, an oral treatment for type 3 Gaucher disease. This rare disease is caused by an enzyme deficiency leading to lysosomal storage, with progressive neurological symptoms. The designation is based on results from the Phase 3 LEAP2MONO study, which demonstrated an improvement in neurological symptoms in patients treated with venglustat compared to standard enzyme replacement therapy.
Sanofi emphasizes that venglustat has not revealed any new significant side effects compared to previous studies. Commonly observed adverse effects include headaches and nausea. Already designated a "fast-track" and "orphan drug," venglustat continues its development, highlighting the need for innovative solutions for neurological symptoms associated with MG3.
R. E.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all SANOFI-AVENTIS news
