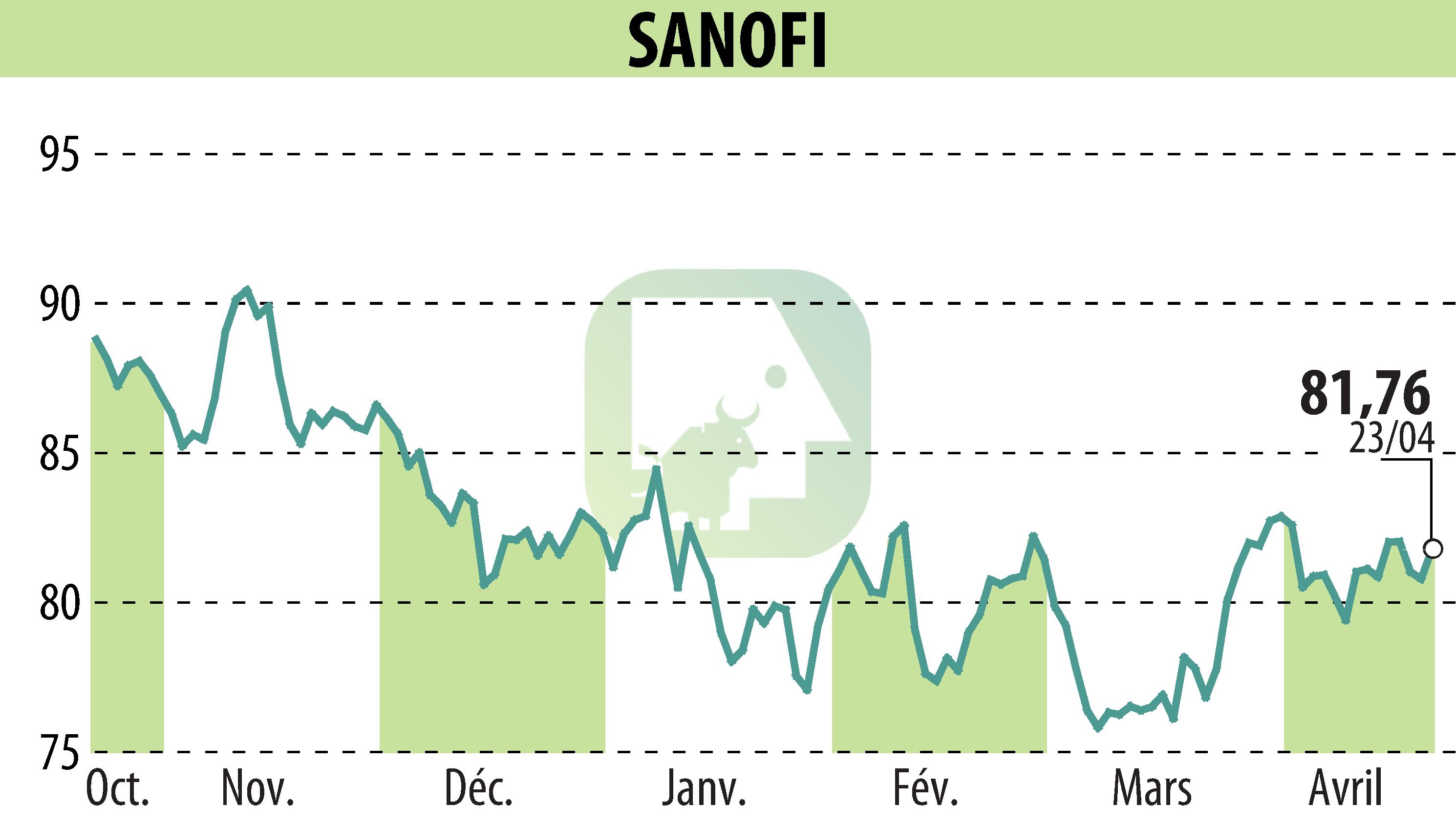
on SANOFI-AVENTIS (EPA:SAN)
Sanofi receives a favorable opinion for tolebrutinib in the treatment of multiple sclerosis
The Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency has recommended the approval of Sanofi's Cenrifki (tolebrutinib). This treatment is intended for secondary progressive multiple sclerosis (SPMS) without relapses. This recommendation is based on the phase 3 HERCULES trial, which demonstrated a significant reduction in disability progression.
MS-SP is a debilitating form of multiple sclerosis characterized by a continuous accumulation of disability. Clinical studies show that tolebrutinib, by penetrating the brain, could slow this progression. However, risks of liver damage have been observed, requiring close monitoring.
R. H.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all SANOFI-AVENTIS news
