on SANOFI-AVENTIS (EPA:SAN)
Sanofi's Cenrifki Receives Positive CHMP Opinion for SPMS
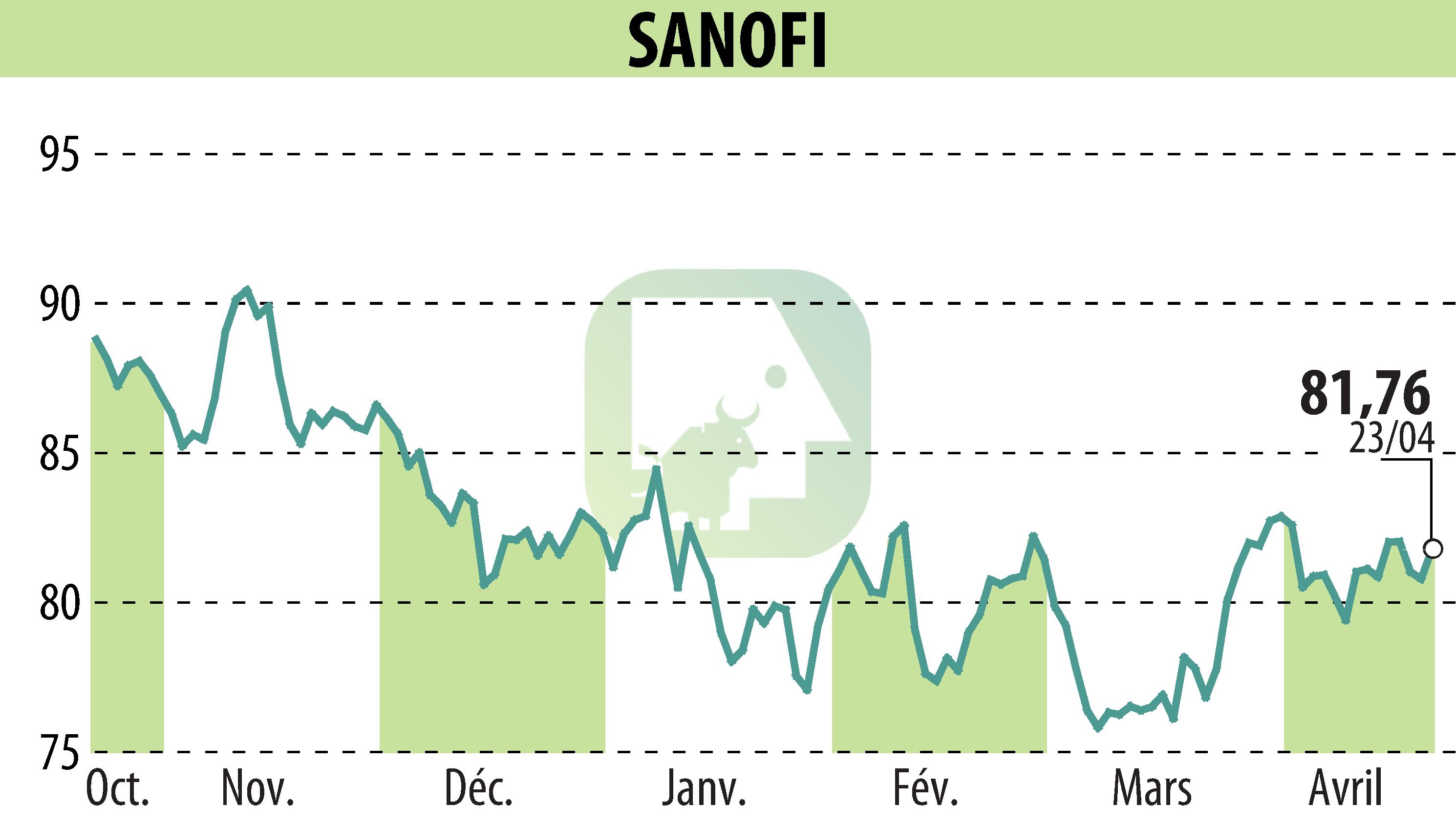
Sanofi has announced that its Bruton’s tyrosine kinase inhibitor, Cenrifki (tolebrutinib), has received a positive opinion from the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP). This recommendation concerns the treatment of secondary progressive multiple sclerosis (SPMS) without relapses. It is based on the results of the HERCULES phase 3 study, which showed a delay in disability progression.
The HERCULES study underlined the significant unmet need in managing legal disabilities affecting SPMS patients. Data from this study, along with GEMINI 1 and 2 studies focused on relapsing multiple sclerosis, supported the CHMP's recommendation.
While Cenrifki's safety profile was consistent, significant liver enzyme elevations were noted, underscoring the need for careful liver monitoring. The final decision from regulatory authorities is expected soon, with global submissions underway.
R. E.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all SANOFI-AVENTIS news
