on SENSORION (EPA:ALSEN)
Sensorion continues its development in gene therapy for deafness
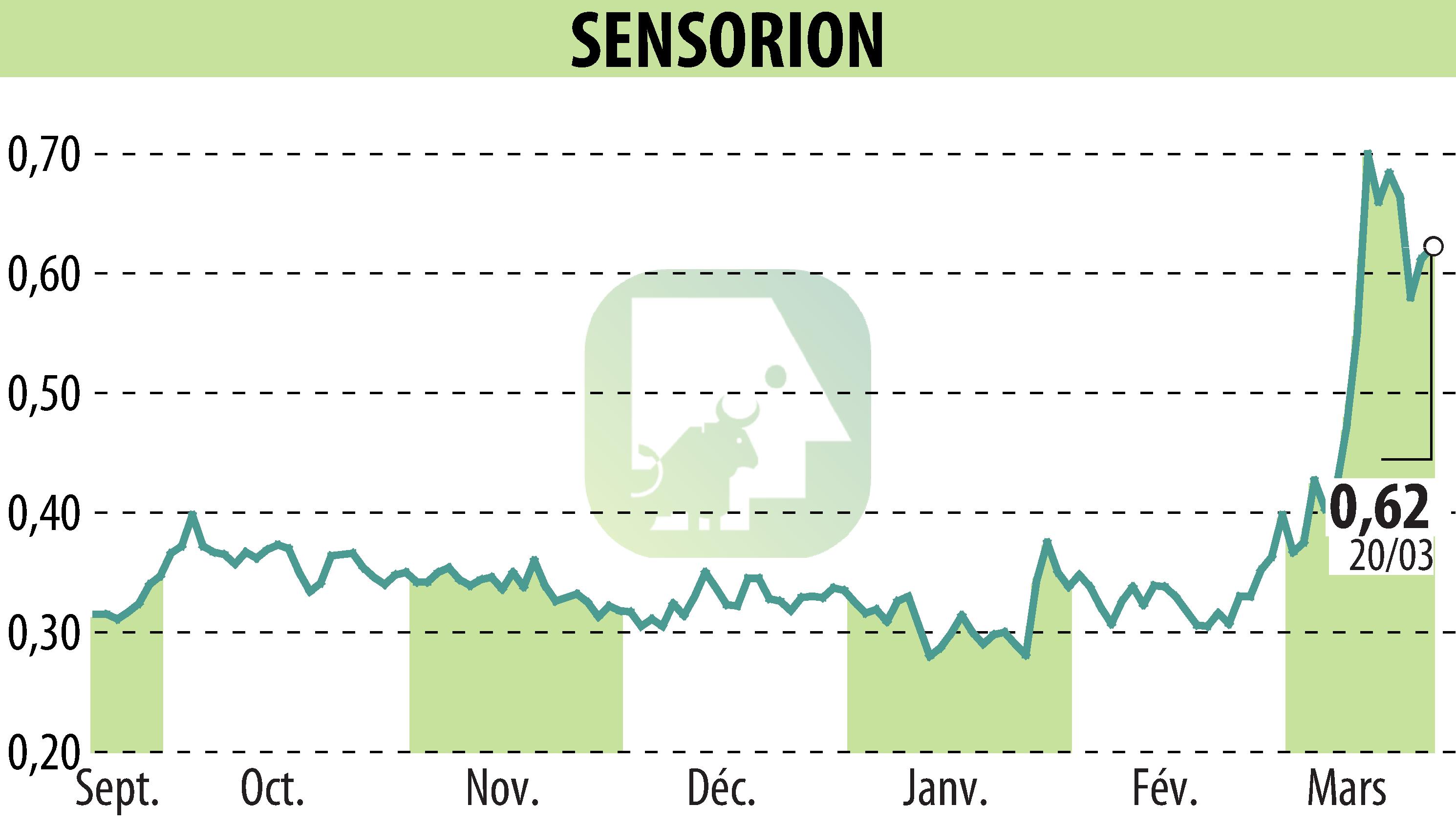
Sensorion has published the six-month results of the Phase 1/2 Audiogene trial of its gene therapy SENS-501. This study targets congenital deafness linked to mutations in the OTOF gene. The data show early signs of efficacy in Cohort 2, suggesting a dose-response relationship. A third dose level is being considered, and consultations with regulatory authorities are planned.
In parallel, Sensorion is making progress on the GJB2-GT (SENS-601) program, which targets deafness linked to the GJB2 gene, the leading cause of congenital deafness. Submission of the CTA is expected in the first half of 2026, and that of the IND by the end of 2026.
Clinical efforts and acquired experience strengthen Sensorion's position in therapeutic innovation for genetic hearing loss, particularly affecting children. This development is supported by strategic collaborations, notably with the Pasteur Institute.
R. P.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all SENSORION news
