on SENSORION (EPA:ALSEN)
Sensorion Updates on Audiogene Trial and Advances GJB2-GT
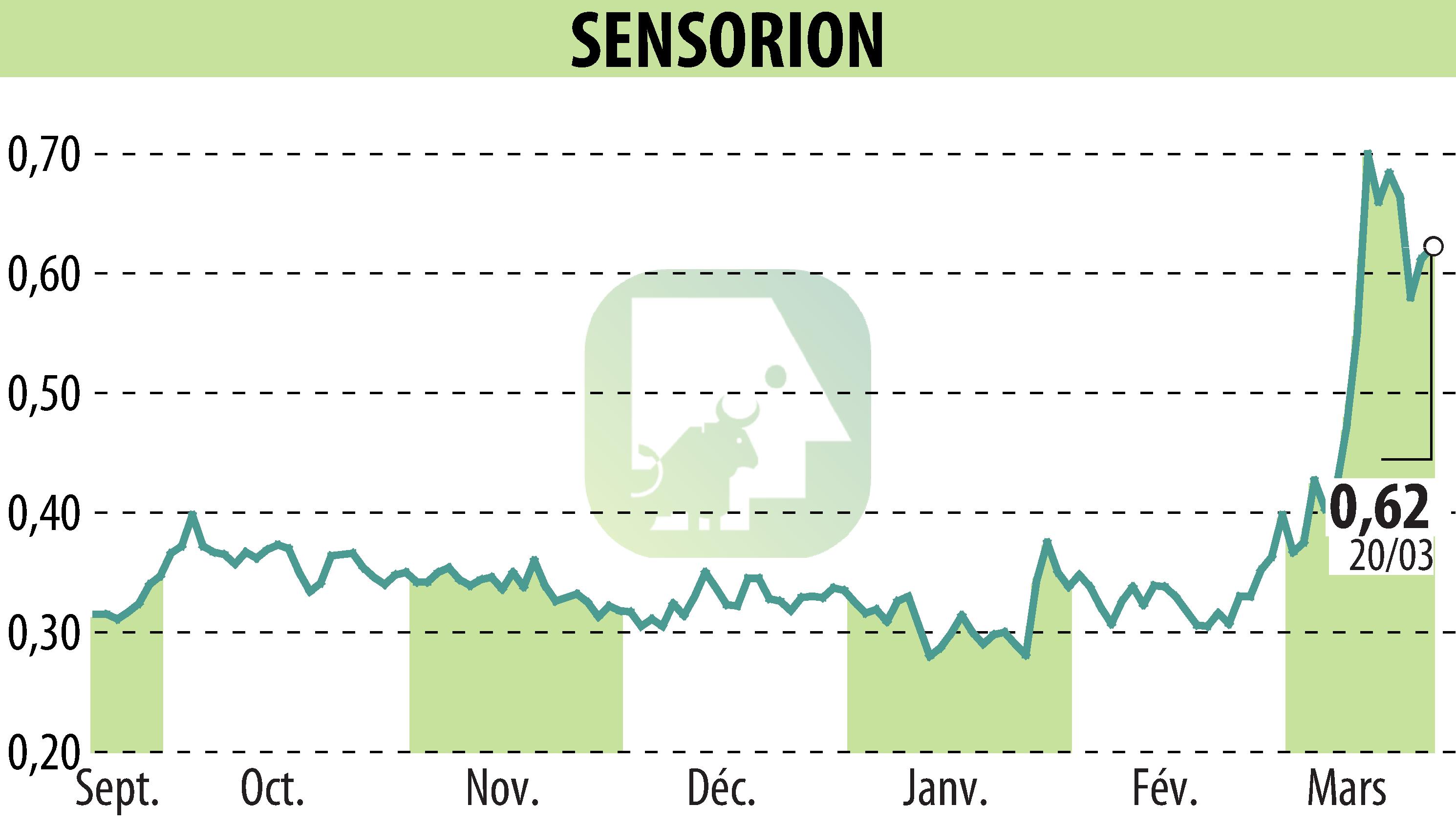
Sensorion, a biotechnology company focused on hearing loss therapies, has released a six-month update from the Audiogene Phase 1/2 trial. The data indicates sustained efficacy of SENS-501 in patients with otoferlin-mediated deafness. A third dose level is being considered, pending regulatory consultations. The trial has shown no serious adverse effects, supporting Sensorion's commitment to gene therapy for hearing loss.
In parallel, Sensorion advances its GJB2-GT (SENS-601) program, targeting genetic deafness caused by GJB2 mutations. Preparations for clinical trials in Europe and the U.S. are underway, with submissions expected by the end of 2026. The program will be showcased at the ASGCT meeting in May 2026.
These developments bolster Sensorion's growing portfolio in gene therapy, aiming to provide new treatments for hearing disorders. Financial backing from strategic investors like Sanofi enhances the company's capability to progress these initiatives.
R. E.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all SENSORION news
